About quantum biology
Quantum biology is the study of why quantum effects may be integral to how biological systems function.
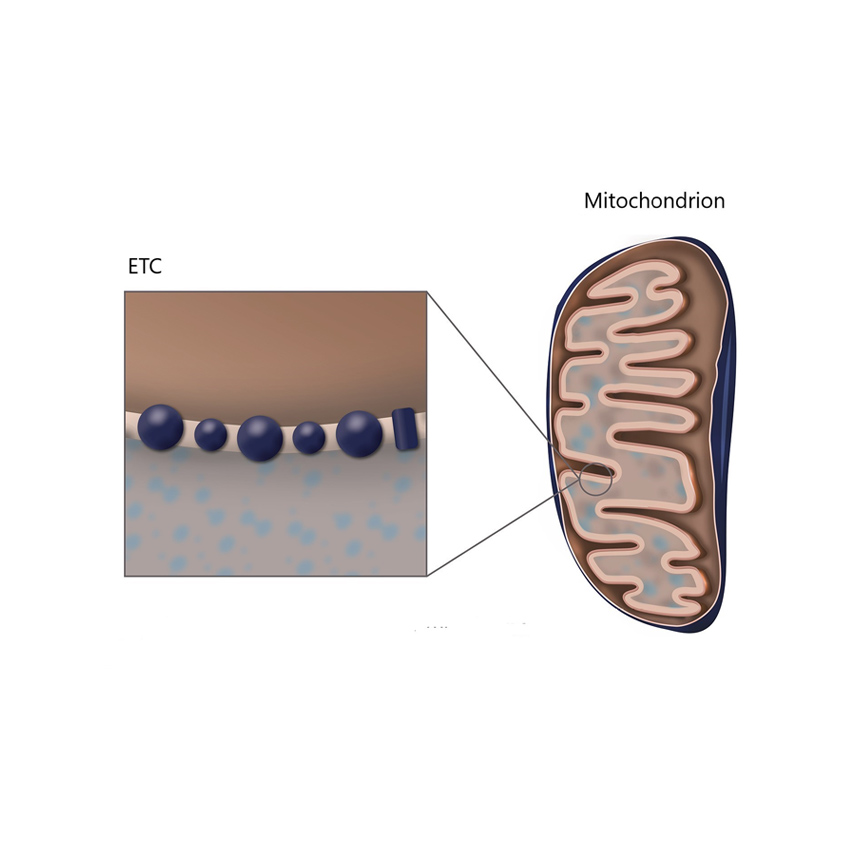
Quantum mechanics is the best theory we have to explain the fundamental behaviour of the universe at the atomic and molecular level, for very short time and length scales. A key principle in quantum theory is that energy is quantised, and that both matter and radiation display wave-particle duality. As our systems of interest increase in size from the atomic, to collections of molecules such as a protein, there is an increasing likelihood that they can be described as particles and behave as such – following the classical laws of Newtonian physics. However, it has been shown that even very large molecules, consisting of thousands of atoms, can exhibit wave-like properties.
The following books provide a useful overview and more detailed discussion of quantum biology:
The Guy Foundation’s Founder and Chairman Professor Geoffrey Guy wrote Quantum Biology as a book on quantum biology for the general reader, but with a scope and depth that will satisfy many scientists and medical professionals. He examines how quantum phenomena affect processes as diverse as the functioning of the brain and the body's approach to combating inflammation and disease as well as how we may need to rethink the ways in which energy and information are carried round our nervous system. Ultimately the book gives an intriguing glimpse into the future of medicine.
The Physical Principles of Quantum Biology book, released in early 2025, was written by Dr Nathan Babcock. It was motivated by the need to assist researchers, especially those entering the field, to have access to a coherent overview of quantum biology.
Visit our Publications and Useful resources pages for further reading, the Glossary for descriptions of key terms, and for talks by a number of scientists working in quantum biology and related fields visit Our online lectures.